

A recent study by UN has confirmed that the ozone layer that shields life from cancer-causing solar rays is recovering at a rate of one to three percent per decade, reversing years of dangerous depletion caused by the release of harmful chemicals.
Context
- A recent study by UN has confirmed that the ozone layer that shields life from cancer-causing solar rays is recovering at a rate of one to three percent per decade, reversing years of dangerous depletion caused by the release of harmful chemicals.
- Ozone is a molecule comprised of three oxygen atoms that occurs naturally in small amounts.
About
- Earth's ozone layer acts like a sunscreen, shielding the planet from potentially harmful ultraviolet (UV) radiation that can cause skin cancer, cataracts, suppress immune systems and damage wildlife. It is produced in tropical latitudes and distributed around the globe.
- The four-yearly review of the Montreal Protocol, a 1987 ban on man-made gases that damage the fragile high-altitude ozone layer, found long-term decreases in the atmospheric abundance of controlled ozone-depleting substances and the on-going recovery of stratospheric ozone.
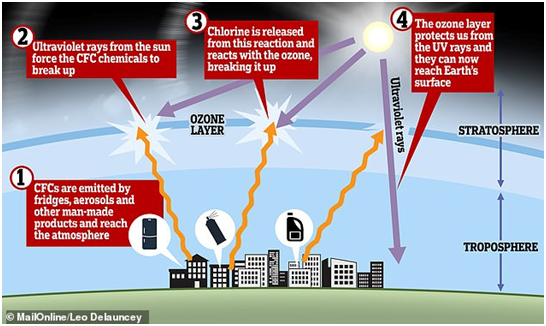
- A hole was found in the ozone layer above Antarctica in the 1980s as a result of harmful chemicals being pumped into the atmosphere. This has allowed large amounts of ultraviolet light to reach Earth unabated and has been linked to an increase in skin cancer diagnoses.
- Scientists have now found that severe bans on the manufacture of these products have seen the hole in the ozone recover at a rate of one to three per cent per decade.
- The U.N. had already hailed the success of the Montreal Protocol, which banned or phased out ozone depleting chemicals, including chlorofluorocarbons (CFCs) once widely used in refrigerators and spray cans and were found to be the main reason behind the loss of the ozone layer, but the report said it was the first time that there were emerging indications that the Antarctic ozone hole had diminished in size and depth since 2000.
Chlorofluorocarbons (CFCs)
- Chlorofluorocarbons are nontoxic, non-flammable chemicals containing atoms of carbon, chlorine, and fluorine. They are used in the manufacture of aerosol sprays, blowing agents for foams and packing materials, as solvents, and as refrigerants.
- CFCs are classified as halocarbons, a class of compounds that contain atoms of carbon and halogen atoms. Individual CFC molecules are labelled with a unique numbering system. For example, the CFC number of 11 indicates the number of atoms of carbon, hydrogen, fluorine, and chlorine.
- Whereas CFCs are safe to use in most applications and are inert in the lower atmosphere, they do undergo significant reaction in the upper atmosphere or stratosphere where they cause damage.
- The study also looked at the thinning of the ozone layer in the Arctic and found the annual variations were much larger. This made it harder for the researchers to confirm if there had been a definite recovery in the layer since 2000.
- However, while most of the banned gases have been phased out, the report found at least one violation of the protocol: an unexpected increase in production and emissions of CFC-11 from eastern Asia since 2012. The report said the source country or countries had not yet been identified.
- If CFC-11 emissions continued at the same rate, return of mid-latitude and polar ozone-depleting chemicals to their 1980 values would be delayed by about 7 and 20 years, respectively.
- The Antarctic ozone hole was expected to gradually close, returning to 1980 levels in the 2060s.
Significance
- The report said it was the first time the Antarctic ozone hole had shrunk in size and depth since 2000.
- At projected rates, Northern Hemisphere and mid-latitude ozone is scheduled to heal completely by the 2030s followed by the Southern Hemisphere in the 2050s and Polar Regions by 2060.


