

Context
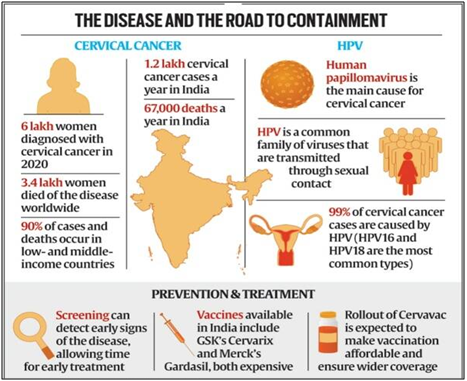
India in the next few months will have an indigenously developed human papilloma virus (HPV) vaccine “Cervavac” that will offer protection against cervical cancer, the second largest killer among cancers in women.
About
What is human papillomavirus infection?
- Human papillomavirus (HPV) is a viral infection that is passed between people through skin-to-skin contact.
- HPV is the most common sexually transmitted infection (STI).
Instances of Infection:
- Almost all cervical cancers are found in women who have a history of HPV infection.
- But having an HPV infection may not lead to cancer in all cases, rather it must be noted that over 80% of women have an HPV infection in their lifetime and almost all of them can clear it.
- But in some people, some high-risk types of HPV persist and lead to cervical cancer.
- In addition, having multiple sexual partners, poor hygiene, and smoking are other factors that increase the risk of developing cervical cancer.
How does vaccination against an infection protect against cancer?
- The best way to prevent cancer is to prevent an infection in the first place and vaccines help to a great extent.
- There are two to four types of HPV that lead to almost 70% to 80% of all cervical cancers and the vaccines prevent these infections. Since the virus is sexually transmitted, it has to be given to teenage girls before they are sexually active.
- Once an infection happens, the vaccine cannot clear it out. Hence, the effectiveness of the vaccine keeps dropping with age.
|
About Cervical Cancer:
|
HPV vaccines presently available in India:
- At present, there are two HPV vaccines available- GSKs Cervarix (which protects against the two most common types HPV 16 and 18) and Merck’s Gardasil (which protects against two additional types 6 and 11).
- These vaccines cost between Rs 2,500 and Rs 3,300 per dose, with teenagers requiring at least two doses and those getting it later in life needing three.
- Drugs Controller General of India (DCGI) has granted market authorization to the Pune-based Serum Institute of India (SII) for the first Quadrivalent Human Papillomavirus vaccine (qHPV) against cervical cancer.
| The quadrivalent human papillomavirus (HPV) vaccine protects against infection with HPV types 6, 11, 16, and 18, which are responsible for 70% to 80% of cervical cancers and at least 90% of cases of genital warts. |
Constraints in Immunization:
- Universal immunization of all teenage girls becomes an expensive proposition.
- Cervarix is not readily available in India so we had to start using the other one, which is costlier.
Significance of India’s first quadrivalent HPV vaccine:
- It will drastically bring down the incidence and deaths caused by cervical cancer in India (India contributes to about a fourth of the global mortality from cervical cancer).
- Once India-made vaccines are available, it will make it cheaper and more cost-effective for governments to give the shot under public health programs. And, hopefully, it will become a part of the national program as well.
Options left to those who missed vaccination:
- Regular screening is the best way to keep an eye out for whether they have cervical cancer.
- HPV test has a higher detection rate in comparison to Pap Smear Test (a procedure where a small brush is used to remove cells from the cervix to be studied under a microscope). The sample is collected similarly but it looks for the high-risk types of HPV in the cells.
- This test should be done starting two to three years after sexual activity begins, then every five years till the age of 65 years.


