

Recent images sent by Chandrayaan-1, India's first lunar mission, suggest that the Moon may be rusting along the poles.
Context
Recent images sent by Chandrayaan-1, India's first lunar mission, suggest that the Moon may be rusting along the poles.
Background
- ISRO's maiden mission to the Moon has sent images which show that the Moon may be rusting along the poles.
- Chandrayaan-1 was launched in 2008.
- The sign of this finding is that even though the surface of the Moon is known to have iron-rich rocks, it is not known for the presence of water and oxygen, which are the two elements needed to interact with iron to create rust.
- The Chandrayaan-1 Moon data indicates that the Moon's poles are home to water, this is what the scientists are trying to decipher.
|
Chandrayaan-1
|
Analysis
Important details about Moon
- The Moon (or Luna) is the Earth’sonly natural satellite and was formed 4.6 billion years ago around some 30–50 million years after the formation of the solar system.
- The Moon is in synchronous rotation with Earth meaning the same side is always facing the Earth.
- The first uncrewed mission to the Moon was in 1959 by the Soviet Lunar Program with the first crewed landing being Apollo 11 in 1969.
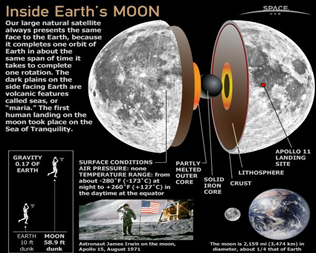
- Atmosphere: The moon has a very thin atmosphere, so a layer of dust — or a footprint — can sit undisturbed for centuries.
- And without much of an atmosphere, heat is not held near the surface, so temperatures vary wildly. Daytime temperatures on the sunny side of the moon reach 273 degrees F (134 C); on the dark side it gets as cold as minus 243 F (minus 153 C).
- The lack of atmosphere means no sound can be heard on the Moon, and the sky always appears black.
|
Orbital characteristics |
|
|
Average distance from Earth |
238,855 miles (384,400 km) |
|
Perigee (closest approach to Earth) |
225,700 miles (363,300 km) |
|
Apogee (farthest distance from Earth) |
252,000 miles (405,500 km) |
|
Orbit circumference |
1,499,618.58 miles (2,413,402 km) |
|
Mean orbit velocity |
2,287 mph (3,680.5 km/h) |
Analysing the findings
- Data from the Indian Space Research Organisation (ISRO) has revealed that the Earth’s natural satellite Moon might be rusting.
- The new research suggests that the moon is turning slightly red, indicating the formation of a reddish-black mineral form of iron named hematite on its surface, particularly at the poles.
- The formation of rust or iron oxide can be attributed to the presence of two key elements—water and oxygen—when they come in contact with iron.
- The lunar surface is littered with iron-rich rocks, which may facilitate this chemical reaction when combined with the other two elements.
- However, the Moon does not have any rich source of water and is devoid of oxygen in its atmosphere.
- It is believed that though Moon lacks atmosphere to support the formation of oxygen, it hosts traces of oxygen that travels from Earth to reach the lunar environment.
What NASA says?
- While the Moon is airless, research indicates the presence of hematite, a form of rust that normally requires oxygen and water. That has scientists puzzled.
- Scientists at the National Aeronautics and Space Administration (NASA) say that this could be because the Earth's atmosphere is lending a helping hand which, in other words, means that the Earth's atmosphere could be protecting the Moon as well.
The rusting chemistry
- Rust is the common name for iron oxide. The most familiar form of rust is the reddish coating that forms flakes on iron and steel (Fe2O3), but rust also comes in other colors including yellow, brown, orange, and even green
- For iron to become iron oxide, three things are required:
- Iron
- Water
- Oxygen
- Rust forms when iron or its alloys are exposed to moist air. The oxygen and water in air react with the metal to form the hydrated oxide.
iron + water + oxygen → hydrated iron(III) oxide
Terrestrial oxygen travels to the moon
- In order to turn iron into rust, an 'oxidizer' is needed. An oxidizer is a molecule such as oxygen that removes electrons from a material such as iron.
- The sun's solar wind, a stream of charged particles that constantly hits the moon with hydrogen, has the opposite effect.
- However, Hydrogen is a reducer or a molecule that provides electrons to other molecules.
- Without protection from this solar wind, such as the magnetic field that shields our planet from it, rust should not be able to form on the moon.
- However it does, and the key might be the earth.
- The moon doesn't have an atmosphere of its own to provide sufficient amounts of oxygen, but it has trace amounts provided by Earth's atmosphere, according to the statement.
- This terrestrial oxygen travels to the moon along with an elongated extension of the planet's magnetic field called a 'magnetotail'.
- Earth's magnetotail can reach all the way to the near side of the moon where more of the hematite was found.
- Furthermore, the magnetotail blocks 99 percent of solar wind from blasting the moon at every full moon, drawing a temporary curtain over the lunar surface, allowing periods of time for rust to form.
What about ‘water’?
- Another ingredient required to form the rust is water and the moon mostly lacks in it.
- The researchers have proposed that fast-moving dust particles that blast the moon might free water molecules locked into the moon's surface layer, allowing the water to mix with the iron.
- These dust particles might even be carrying water molecules themselves, and their impact might create heat that could increase the oxidation rate.
|
Which other planet is rusting?
|
Is Earth to be ‘blamed’?
- For iron to convert into rust it needs oxidizers, which can steal electrons from the iron and thus initiate rust formation.
- To understand the cause, the team examined the bombarding of hydrogen from the solar wind. However, it had the opposite effect since hydrogen is a reducer—which donates electrons instead of taking it.
- Therefore, solar winds may not be the reason behind rusting on the Moon’s surface.
- The airless Moon may lack atmosphere to support the formation of oxygen, but it hosts traces of oxygen that travels from Earth to reach the lunar environment.
- Therefore, researchers say the Earth’s magnetotail—an elongated region of the planet’s magnetosphere—plays a significant role in this change observed over the Moon.
- In 2007, Japan's Kaguya orbiter helped scientists discover that oxygen from Earth's upper atmosphere can travel through magnetotail to the Moon, which is 3,85,00 kilometres away from the Earth.
- Therefore, the terrestrial oxygen can reach the near side of the Moon facing the Earth and this finding means that the oxygen from Earth may be driving the formation of hematite on the lunar surface.
- In addition, the magnetotail interferes with the hydrogen molecules released from the solar wind to reach the Moon atmosphere, which if reached may inhibit this rust formation.
Conclusion
The new findings will reshape our knowledge about the Moon's polar regions. Earth may have played an important role on the evolution of the Moon's surface.


