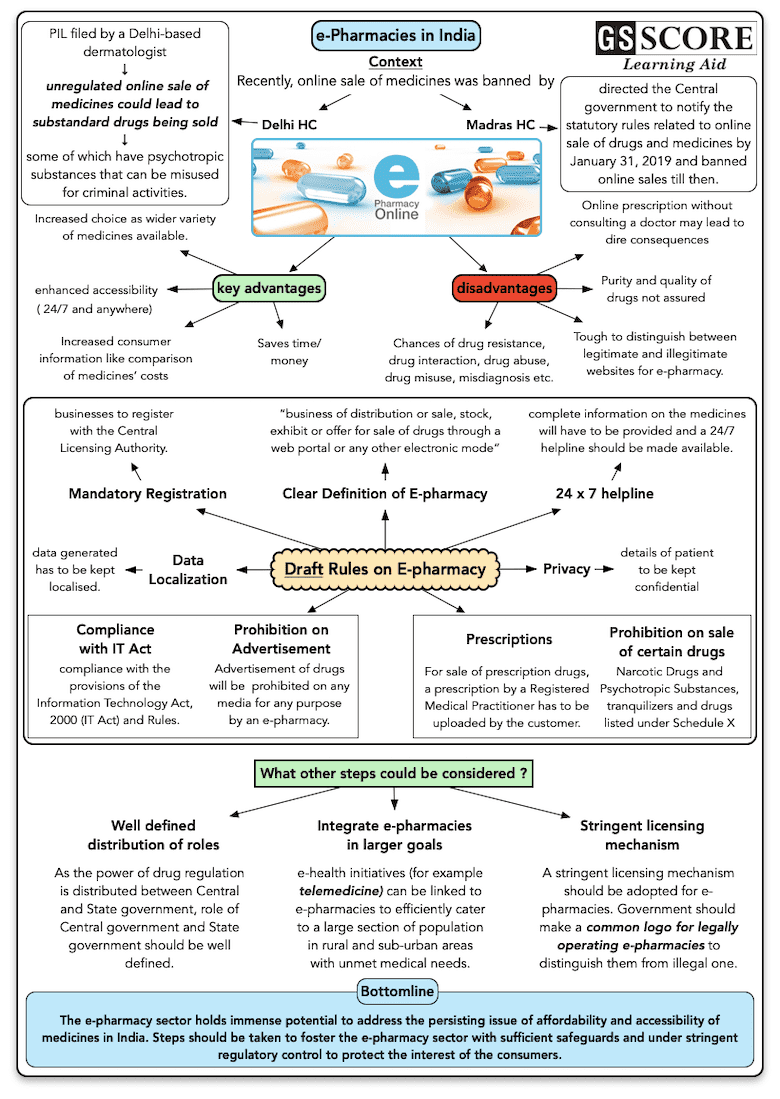
- Recently both Madras and Delhi High Court has banned the online sale of medicines.
- The Madras High Court has directed the Central government to notify the statutory rules related to online sale of drugs and medicines by January 31, 2019 and banned online sales till then.
Issue
Context
- Recently both Madras and Delhi High Court has banned the online sale of medicines.
- The Madras High Court has directed the Central government to notify the statutory rules related to online sale of drugs and medicines by January 31, 2019 and banned online sales till then.
Background
- The Delhi High Court order was in response to a PIL filed which held that unregulated online sale of medicines could lead to substandard drugs being sold, some of which have psychotropic substances that can be misused for criminal activities.
- Madras High Court held that the pharmacies in the country would be entitled to sell medicines online only after obtaining licences under the rules that were now in the draft stage and yet to be finalised.
- As per the Drugs and Cosmetics Rules of 1945, it is not permitted to ship, mail or provide door delivery of the prescribed medicines. Yet, drugs of scheduled and non-scheduled category are available online on different websites and distributed across the country.
About
E-pharmacies
- E-pharmacy denotes the buying and selling of medicines and other pharmaceutical items with the support of web portal or any electronic mode. These are online platforms where consumers can purchase medicines without having to visit brick-and-mortar pharmacies.
- The advent of internet pharmacy and access of drugs and diagnostics is gaining popularity due to cost effectiveness, high speed delivery to door step of patients.
- The Indian pharmacy market is huge and, as of 2017, is the third largest in terms of volume and 13th largest in terms of value, globally. However, the e-pharmacy market is estimated to be a fraction of that. There are more than 200 e-pharmacy start-ups in India.
Analysis
E-Pharmacies regulation in India
- Currently, regulatory powers have been distributed between the centre and the state governments. Central Government is responsible for licensing of drug imports and the state governments are responsible for the manufacture, sale and distribution of drugs.
- Central Government exercises regulatory control over drugs by Central Drugs Control Organisation headed by the Drugs Controller General of India. State authorities’ exercises regulatory control over drugs by state based Drugs Control Administration headed by the State Drugs Controllers. Every state has its own Drugs Control Administration.
- The laws governing pharmacies in India are derived from Drugs and Cosmetics Act, 1940; Drugs and Cosmetics Rules, 1945; Pharmacy Act, 1948; Indian Medical Act, 1956 and Code of Ethics Regulations, 2002, etc.
- These all laws were written before the digital era. Thus, there are no laws related to internet and e-commerce. The Information Technology Act, 2000 governs all activities and issues related to internet.
- E-pharmacies come under the purview of the Drugs and Cosmetics Act, 1940 and the Information Technology Act, 2000. But, existing Drugs and Cosmetics Act, 1940 doesn’t distinguish between online and offline pharmacies.
Advantages
- 24/7 access possible
- Economical with refund possible
- Easy comparison of medicines in terms of cost
- Increased consumer information and information exchange
- Privacy
- Fast distribution
- Increased choice as wider variety of medicines available
- Convenient for some patients and old age people who can’t leave their home
- Delivery of medicines at desired place at desired time possible
Disadvantages
- Chances of drug resistance, drug interaction, drug abuse, drug misuse, misdiagnosis etc.
- Promote self-medication
- Purity and quality of drugs not assured
- Financial privacy issues
- Medical privacy is a major concern
- Electronic health records security and privacy concerns
- Doctor’s prescription may not be honoured
- Affects business of offline pharmacists
- Access to illiterate and poor population difficult
- Tough to distinguish between legitimate and illegitimate websites for e-pharmacy
- No control on purchasing drugs by minors from e-pharmacies
- Tough to transport temperature sensitive drugs
Challenges
- Absence of concrete laws for e-pharmacies in India
- Selling or shipment of drugs to minors
- Reach of technology driven model to illiterate people due to lack of knowledge about internet
- Speed of internet
- Prescription related issues
- Legality of electronic signature
- Identity and reliability of legal e-pharmacy
- Protection of consumer rights
- Security and confidentiality of information exchanged
- Security of financial transactions
- Regulatory control over e-pharmacies operating outside the jurisdiction of India
- Unclear laws on inter-state transfer of drugs/medicines
- Drug importation and re-importation issue
Draft Rules on E-pharmacy
The Union Health Ministry has recently come out with draft rules on sale of drugs by e-pharmacies with an aim to regulate online sale of medicines across India and provide patients accessibility to genuine drugs from authentic online portals. The draft rules on "sale of drugs by e-pharmacy" state that no person will distribute or sell, stock, exhibit or offer for sale of drugs through e-pharmacy portal unless registered.
Key highlights:
- Definition of E-pharmacy: “Business of distribution or sale, stock, exhibit or offer for sale of drugs through a web portal or any other electronic mode”.
- Mandatory Registration: it is mandatory for e-pharmacy businesses to register with the Central Licensing Authority.
- Data Localization: It mandates e-pharmacy portals to be established in India through which they are conducting their business and the data generated has to be kept localised.
- Privacy: It states that the details of patient should be kept confidential and not to be disclosed to any third party except the central government or the state government concerned.
- Prescriptions: For carrying out sale of prescription drugs (i.e. drugs listed under Schedule H, H1 of the Drugs and Cosmetic Rules) a prescription by a Registered Medical Practitioner has to be uploaded by the customer.
- Prohibition on sale of certain drugs: Sale of drugs covered by the Narcotic Drugs and Psychotropic Substances Act, 1985, tranquilizers and drugs listed under Schedule X has been prohibited. Schedule X drugs include narcotics and psychotropic substances.
- Prohibition on Advertisement: Advertisement of drugs is prohibited on any media for any purpose by an e-pharmacy.
- Compliance with IT Act: E-pharmacies have to comply with the provisions of the Information Technology Act, 2000 (IT Act) and Rules.
- 24*7 helpline: The rules state that complete information on the medicines will have to be provided by the e-pharmacy holders, and a 24/7 helpline should be made available.
Way Forward
- The e-pharmacy sector holds immense potential to address the persisting issue of affordability and accessibility of medicines in India. Steps should be taken to foster the e-pharmacy sector with sufficient safeguards and under stringent regulatory control to protect the interest of the consumers.
- Specific and clear-cut rules should be made for selling, prescribing, dispensing, and delivering prescription drugs through e-pharmacies.
- A stringent licensing mechanism should be adopted for e-pharmacies. Government should make a common logo for legally operating e-pharmacies to distinguish them from illegal one. For example: The European Union (EU) has issued a common logo for legally operating e- pharmacies in the EU member states.
- As the power of drug regulation is distributed between Central and State government, role of Central government and State government should be well defined.
- E-pharmacy must establish its server in India as if it is outside the boundaries of India, it is difficult to control and regulate it.
- Government schemes like National Rural Health Mission can aid in promoting proper procedures to acquire drugs, prevent self-medication through campaigns on television, radios and social media.
- e-health initiatives for example telemedicine can be linked to e-pharmacies to efficiently cater to a large section of population in rural and sub-urban areas with unmet medical needs.
Learning Aid
Practice Question:
Critically analyse the status of e-pharmacies in India. Suggest some way forward.