- Recently, a Chinese scientist has claimed to have created the world’s first genetically edited babies using CRISPR-Cas9 technology, in a potentially ground-breaking and controversial medical first.
- If true, it would be a profound leap of science and ethics. This kind of gene editing is banned in most countries as the technology is still experimental and DNA changes can pass to future generations, potentially with unforeseen side-effects.
Issues
Context
- Recently, a Chinese scientist has claimed to have created the world’s first genetically edited babies using CRISPR-Cas9 technology, in a potentially ground-breaking and controversial medical first.
- If true, it would be a profound leap of science and ethics. This kind of gene editing is banned in most countries as the technology is still experimental and DNA changes can pass to future generations, potentially with unforeseen side-effects.
Background
- CRISPRs were first discovered in archaea (and later in bacteria) by Francisco Mojica. He proposed that CRISPRs serve as part of the bacterial immune system, defending against invading viruses. They consist of repeating sequences of genetic code, interrupted by “spacer” sequences – remnants of genetic code from past invaders.
- The system serves as a genetic memory that helps the cell detect and destroy invaders (called “bacteriophage”) when they return.
About
What are Genes and Gene- editing?
- Genes contain the bio-information that defines any individual. Physical attributes like height, skin or hair colour, more subtle features and even behavioural traits can be attributed to information encoded in the genetic material. An ability to alter this information gives scientists the power to control some of these features.
Genome editing
- Genome editing/gene editing, is a group of technologies that give scientists the ability to change an organism's DNA. These technologies allow genetic material to be added, removed, or altered at particular locations in the genome.
- Several approaches to genome editing have been developed. A recent one is known as CRISPR-Cas9, which is short for Clustered Regularly Interspaced Short Palindromic Repeats and CRISPR-associated protein 9.
- The CRISPR-Cas9 system is faster, cheaper, more accurate, and more efficient than other existing genome editing methods.
CRISPR-Cas9
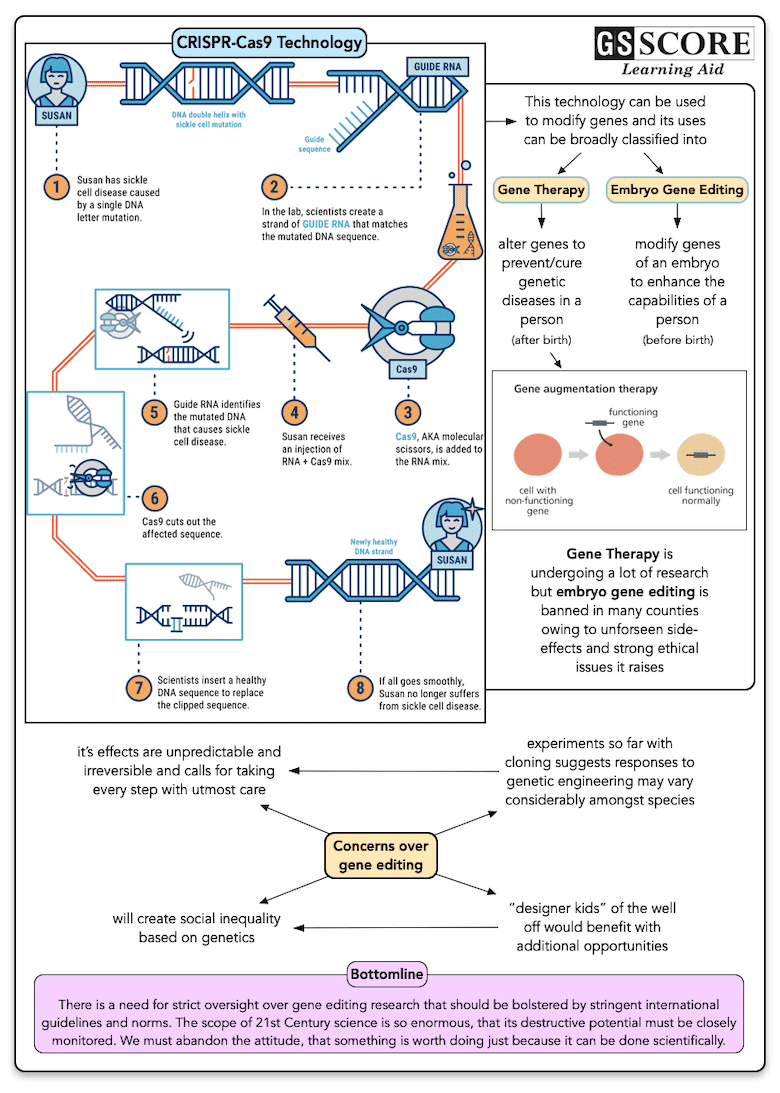
- It is a unique technology that enables geneticists and medical researchers to edit parts of the genome by removing, adding or altering sections of the DNA sequence.
- CRISPRs are specialized stretches of DNA. The protein Cas9 (or "CRISPR-associated") is an enzyme that acts like a pair of molecular scissors, capable of cutting strands of DNA. It allows researchers to easily alter DNA sequences and modify gene function.
- It is the simplest yet powerful tool for editing genomes and also termed as the most versatile and precise method of genetic manipulation.
Analysis
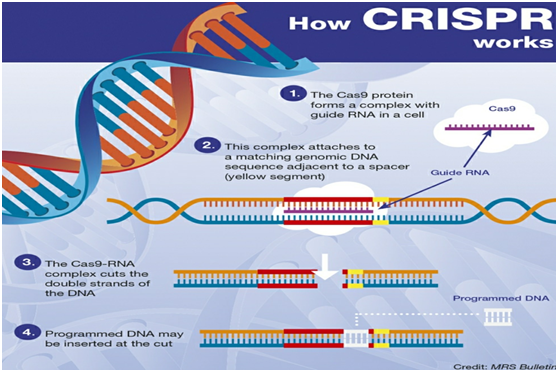
How does it work?
The CRISPR-Cas9 system consists of two key molecules that introduce a change (mutation) into the DNA. These are:
- An enzyme called Cas9. This acts as a pair of ‘molecular scissors’ that can cut the two strands of DNA at a specific location in the genome so that bits of DNA can then be added or removed.
- A piece of RNA called guide RNA (gRNA). This consists of a small piece of pre-designed RNA sequence (about 20 bases long) located within a longer RNA scaffold. The scaffold part binds to DNA and the pre-designed sequence ‘guides’ Cas9 to the right part of the genome. This makes sure that the Cas9 enzyme cuts at the right point in the genome.
- The guide RNA is designed to find and bind to a specific sequence in the DNA. The guide RNA has RNA bases that are complementary to those of the target DNA sequence in the genome. This means that the guide RNA will only bind to the target sequence and no other regions of the genome.
- The Cas9 follows the guide RNA to the same location in the DNA sequence and makes a cut across both strands of the DNA.
- At this stage the cell recognises that the DNA is damaged and tries to repair it.
- Scientists can use the DNA repair machinery to introduce changes to one or more genes in the genome of a cell of interest.
Applications and Implications
- Genome editing is of great interest in the prevention and treatment of human diseases. Currently, most research on genome editing is done to understand diseases using cells and animal models.
- The technology is being explored in research on a wide variety of diseases, including single-gene disorders such as cystic fibrosis, haemophilia, and sickle cell disease.
- It also holds promise for the treatment and prevention of more complex diseases, such as cancer, heart disease, mental illness, and human immunodeficiency virus (HIV) infection.
- CRISPR-Cas9 has a lot of potential as a tool for treating a range of medical conditions that have a genetic component, including cancer, hepatitis B or even high cholesterol.
- Many of the proposed applications involve editing the genomes of somatic (non-reproductive) cells but there has been a lot of interest and debate about the potential to edit germline (reproductive) cells.
Implications
The Ethical dilemma
- Making genetic modifications to human embryos and reproductive cells such as sperm and eggs is known as germline editing. Since any changes made in germline cells using CRISPR-Cas9 technology will be passed on from generation to generation. It has raised a number of ethical concerns.
- Another ethical challenge germline cell and embryo genome editing brings up includes whether it would be permissible to use this technology to enhance normal human traits (such as height or intelligence).
- There are growing concerns of trying to produce “designer” babies or altered human beings.
- Based on concerns about ethics and safety, germline cell and embryo genome editing are currently illegal in many countries.
Other Concerns
- Variable efficacy: CRISPR technology is not hundred percent efficient. The genome-editing efficiencies can vary.
- Off-target effects and imprecise edits: There is also the phenomenon of "off-target effects," where DNA is cut at sites other than the intended target. This can lead to the introduction of unintended mutations. Furthermore, even when the system cuts on target, there is a chance of not getting a precise edit. This is called as "genome vandalism."
- There is also a potential ecological impact of using gene drives. An introduced trait could spread beyond the target population to other organisms through crossbreeding. Gene drives could also reduce the genetic diversity of the target population.
Way Forward
- Experts recommend that germline editing should be done only on genes that lead to serious diseases and when no other reasonable treatment alternatives exist.
- Among other criteria, they stress the need to have data on the health risks and benefits and the need for continuous oversight during clinical trials. They also recommend following up on families for multiple generations.
- CRISPR technology is indeed a path-breaking technology, to alter genes in order to tackle a number of conventional and unconventional problems. The most promising use of the CRISPR technology is in treatment of diseases. For example, sickle cell anaemia.
- However, experiments and tests to validate its use must be subjected to appropriate scrutiny by the regulators, and their use must be controlled to prevent commercial misuse.
- Scientists across the world are still working to determine whether the CRISPR technology is safe and effective for use in people.
Learning Aid
Practice Question:
What do you understand by CRISPR Cas9 editing technology? Recently there have been growing ethical concerns regarding the genome editing technology. Discuss. Also, enumerate possible applications of the technology.