

Context
Recently, the Genetic Engineering Appraisal Committee (GEAC) under the Union Ministry of Environment, Forest and Climate Change recommended the “environmental release” of the transgenic hybrid mustard DMH-11 for seed production and ordered to conduct the field demonstration studies with respect to its effects on honey bees and other pollinating insects.
Let us understand exactly about the GM Mustard crops in India.
About
What is Hybridisation of crops?
- Hybridisation involves crossing two genetically dissimilar plant varieties that can even be from the same species.
- The first-generation (F1) offspring from such crosses tend to have higher yields than what either parent can individually give.
Is it possible to produce hybrids of all crops?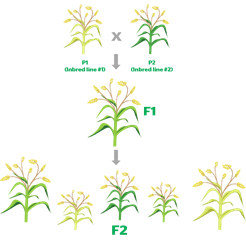
- Hybridisation is not easy for all crops including mustard, as its flowers have both female (pistil) and male (stamen) reproductive organs, making the plants largely self-pollinating.
- Since the eggs of one plant cannot be fertilised by the pollen grains from another, it limits the scope for developing hybrids — unlike in cotton, maize or tomato, where this can be done through physical removal of anthers.
|
The barnase-barstar system enables breeding of hybrids from a wider range of mustards, including those of East European origin such as ‘Heera’ and ‘Donskaja’. |
How has hybridisation been achieved in mustard?
- The hybrid mustard DMH-11 has been produced which contains two alien genes isolated from a soil bacterium called Bacillus ‘amyloliquefaciens’.
- The first gene (‘barnase’) codes for a protein that impairs pollen production and renders the plant into which it is incorporated male-sterile.
- This plant is then crossed with a fertile parental line containing, in turn, the second ‘barstar’ gene that blocks the action of the barnase gene.
- The resultant F1 progeny is both high-yielding and also capable of producing seed/ grain.
- This system was used to develop DMH-11 by crossing a popular Indian mustard variety ‘Varuna’ (the barnase line) with an East European ‘Early Heera-2’ mutant (barstar).
|
Significance: DMH-11 is claimed to have shown an average 28% yield increase over Varuna in contained field trials carried out by the Indian Council of Agricultural Research (ICAR). |
Role of GEAC:
- GEAC is a body responsible for appraisal of proposals relating to the release of GM organisms and products (ordinarily considered hazardous) into the environment.
- The GEAC is the apex biotech regulatory body in India. It is a statutory body.
- The functions of the GEAC are:
- It is responsible for the appraisal of activities that involve the large scale use of hazardous microbes and recombinants in research and industrial production from the point of view of the environment.
- The GEAC also assesses proposals regarding the release of genetically engineered products and organisms into the environment, and this includes experimental field trials as well.
- The body also looks into proposals regarding the use of living modified organism that comes in the risk category III and above in the import/manufacture of recombinant pharma products, or where the end-product of the recombinant pharma product is a modified living organism.
- The Committee has the power to take punitive action against people/body under the Environment (Protection) Act.
- The approval of the GEAC is mandatory before genetically modified organisms and products derived from them can be used commercially.
Concerns associated:
- The presence of a third ‘bar’ gene, which makes GM mustard plants tolerant to the spraying of glufosinate ammonium, a chemical used for killing weeds. This will cause displacement of manual labour engaged in weeding by promoting use of chemical herbicides.
- The GM mustard can threaten the population of honey bees as the Mustard flowers are a source of nectar for honey bees and many other pollinator insects.


