

5th October 2023 (9 Topics)
Context
Moungi G. Bawendi, Louis E. Brus and Alexei I. Ekimov trio has been awarded the Nobel Prize in Chemistry 2023 for the discovery and development of quantum dots.

What are quantum dots?
- Quantum dots (QDs) are man-made nanoscale crystals that exhibit unique optical and electronic properties, including the ability to transport electrons and emit light of various colors when exposed to UV light.
- These artificially synthesized semiconductor nanoparticles have a wide range of potential applications, including use in composites, solar cells, fluorescent biological labeling, displays, lighting, and medical imaging.
- They were first discovered in 1980.
The discovery:
- Their study revealed that, Electrons can absorb energy and emit light of a certain colour, depending on the size of the quantum dot.
- When semiconductor particles are made small enough, they exhibit quantum effects, which restrict the energies at which electrons and holes (the absence of electrons) can exist within the particle.
- As energy is linked to wavelength (or color), this results in the optical properties of the particle being tunable based on its size.
- By controlling the size of the particle, it can be made to emit or absorb specific wavelengths (colors) of light.
About contributions:
- Alexei Ekimov maps the mysteries of coloured glass, studying optical methods, used as diagnostic tools for assessing the quality of semiconducting material. Researchers shine light on the material and measure the absorbance.
- This reveals what substances the material is made from and how well-ordered the crystal structure is.
- Louis E. Brus shows that the strange properties of particles are quantum effects: This was the first time someone had succeeded in deliberately producing quantum dots – nanoparticles that cause size-dependent quantum effects.
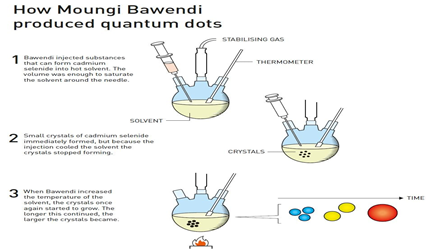
- Moungi Bawendi and his research group succeeded in growing nanocrystals of a specific size. During this phase, the solvent helped give the crystals a smooth and even surface.
- The nanocrystals that Bawendi produced were almost perfect, giving rise to distinct quantum effects. Because the production method was easy to use, it was revolutionary – more and more chemists started working with nanotechnology and began to investigate the unique properties of quantum dots.
Application and Uses of Quantum dots:
- In Television: The luminous properties of quantum dots are utilised in computer and television screens based on QLED technology, where the Q stands for quantum dot.
- In these screens, blue light is generated using the energy-efficient diodes that were recognised with the Nobel Prize in Physics 2014.
- Quantum dots are used to change the colour of some of the blue light, transforming it into red or green.
- This makes it possible to produce the three primary colours of light needed in a television screen.
- In LED lamps: Similarly, quantum dots are used in some LED lamps to adjust the cold light of the diodes.
- The light can then become as energising as daylight or as calming as the warm glow from a dimmed bulb. The light from quantum dots can also be used in biochemistry and medicine.
- In Biochemistry: Biochemists attach quantum dots to biomolecules to map cells and organs.
- Doctors have begun investigating the potential use of quantum dots to track tumour tissue in the body.
- Chemists instead use the catalytic properties of quantum dots to drive chemical reactions.
More Articles


